A research team led by Lawrence Berkeley National Laboratory (Berkeley Lab) and UC Berkeley has engineered bacteria to produce new-to-nature carbon products that could provide a powerful route to sustainable biochemicals, offering sustainable alternatives to chemical manufacturing processes typically reliant on fossil fuels.
They presented their paper in the journal Nature.
The team used bacteria to combine natural enzymatic reactions with a new reaction called the “carbene transfer reaction.” Carbenes are highly reactive carbon-based chemicals scientists have wanted to use for fuel manufacturing, drug discovery, and synthesis. Until now, these carbene processes could only happen in small, test-tube batches, which required expensive chemicals to drive the reaction.
In their study, the team replaced those expensive chemical reactants with natural products produced by the Streptomyces bacteria. The bacteria uses sugar to produce chemical products through cellular metabolism—a much more environmentally friendly process than those used today.
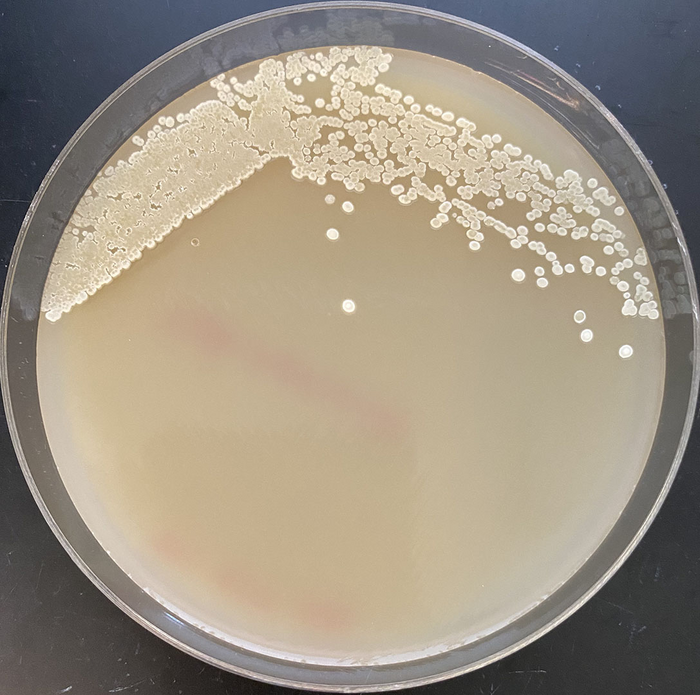
CREDIT
Jing Huang/Berkeley Lab
As the engineered bacterium metabolized and converted sugars into carbene precursors and alkene substrate, it also expressed an evolved P450 enzyme that used those chemicals to produce cyclopropanes, high-energy molecules that can potentially be used in the sustainable production of bioactive compounds and advanced biofuels.
Using this bacterial process to synthesize chemicals can play an integral role in reducing carbon emissions, as the production of chemicals, iron, steel, and cement accounts for nearly 50% of greenhouse gas emissions.
“We can now perform these interesting reactions inside the bacterial cell. The cells produce all of the reagents and the cofactors, which means that you can scale this reaction to very large scales” for mass manufacturing, said Jay Keasling, a principal investigator of the study and CEO of the Department of Energy’s Joint BioEnergy Institute (JBEI).
“This work enables us to perform the carbene chemistry without toxic solvents or toxic gases typically used in chemical synthesis,” said first author Jing Huang, a Berkeley Lab postdoctoral researcher in the Keasling Lab. “This biological process is much more environmentally friendly than the way chemicals are synthesized today.”