Interlocking daisy-chain-like molecular structures that can switch from an expanded and contracted position based on the removal and addition of zinc, mimicking muscle behavior have been made by scientists in Taiwan. Their work appears in Nature Chemistry. Molecular switches have been of interest to scientists for many years. One design for molecular switches is through interlocking molecules, known as rotaxanes, in which a linear molecule is threaded into a macrocycle. The interlocked molecule can selectively encircle different positions on the linear thread when an external stimulus is applied.
In the current study Chang, et al. made monomers that have both a linear component and a macrocyclic component on the same molecule with the idea that in solution these monomers would assemble in a daisy-chain-like fashion. To control how many monomers aggregated, they varied the spacer between the bipyridine portion on the straight arm and the 2,6-bis(hydroxymethyl)pyridine portion of the macrocycle. This allowed them to tailor whether the supramolecular structure favored two ([c2]DC), three ([c3]DC), or four ([c4]DC) interwoven monomers.
Using NMR methods, including diffusion-ordered spectroscopy, and x-ray crystallography, they found that when there was no spacer between bipyridine and 2,6-bis(hydroxymethyl)pyridine, then the [c4]DC molecule was favored. When they used acetylene, benzene, or both as a spacer, then the [c3]DC molecule was favored. The [c2]DC molecule was favored when the spacer was a benzene with acetylenes on either side of it.
The trend is that as the length of the spacer increases, the interlocking supramolecular structure goes from favoring [c4]DC to [c2]DC. The authors suggest that this is due to decreasing the torsional strain, and as the molecules become more flexible, entropy becomes a more dominant factor for which product is favored.
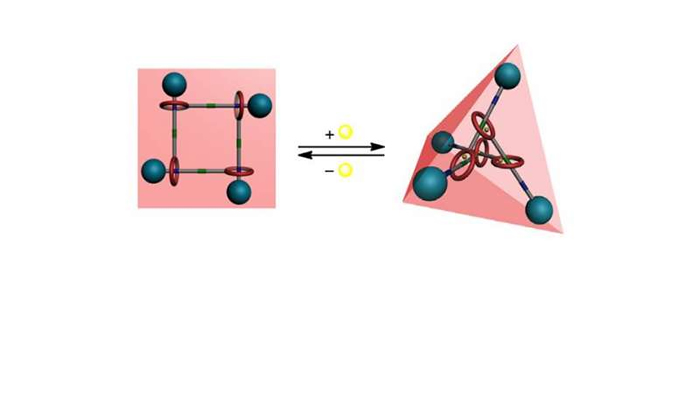
Daisy chains were assembled with Zn(ClO4)2. Zinc coordinates to the bipyridine portion on the straight arm and the 2,6-bis(hydroxymethyl)pyridine portion on the macrocycle. The interlocked molecules were capped by reacting the aniline with an isocyanate compound that has benzene with two trifluoromethyl substituents. This creates a urea derivative that serves as the second station of the molecular switch.
When Zn2+ is not present, the macrocycle surrounds the urea component of the monomer. When zinc is added, the macrocycle moves to the bipyridine station, and the entire structure expands. When TPEN, a zinc sequester is added to solution, the monomers migrated back to their original position in which the urea component is encircled by the macrocycle, or the contracted position.
Using NMR, rotating frame Overhauser spectroscopy, and X-ray crystal analysis, Chang, et al. determined that the [c3]DC structure was planar triangular and the [c4]DC structure was planar quadrangular. The addition of Zn2+ resulted in two-dimensional motion for [c3]DC. The [c4]DC compound, on the other hand, exhibited three-dimensional motion. When expanded, it changed from a planar structure to a tetrahedron. Basic calculation showed that [c3]DC expands to a 23% increase in triangle side length. [c4]DC expands to an 36% increase in side length. This can be compared to human muscle which has a percentage increase of 27%.
This research demonstrates the possibility of designing molecular switches that can expand and contract in both two- and three-dimensions. Future research includes exploring other structures as well as tailoring molecular expansion and contraction stimuli.
More information: Phys.org