There’s a new subject matter being studied in the tech world called transient electronics, which involve devices that would be able to perform all sorts of functions until they’re exposed to light, heat or liquid, at which point they would self-destruct.
Self-destructing electronic devices have the potential to keep military secrets out of enemy hands, or even help patients avoid the removal of medical devices. There are other environmental applications for such technology as well in which monitoring sensors could wash away in the rain.

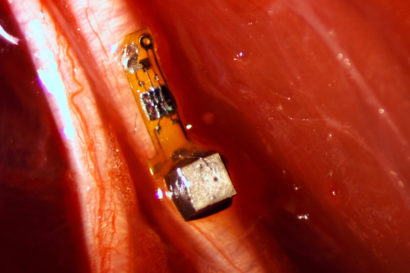
Now, an Assistant Professor of Mechanical Engineering and associate of the U.S. Department of Energy’s Ames Laboratory, Reza Montazami from Iowa State University has developed a lithium-ion battery capable of delivering 2.5 volts and dissolving in just 30 minutes when dropped in water. The batteryis powerful enough to run a desktop calculator for about 15 minutes.
The development is a major breakthrough in the transient electronics realm because it is the first to demonstrate the power, stability and shelf life for practical use in this field.
“Unlike conventional electronics that are designed to last for extensive periods of time, a key and unique attribute of transient electronics is to operate over a typically short and well-defined period, and undergo fast and, ideally, complete self-deconstruction and vanish when transiency is triggered,” the scientists wrote in their paper.
When it comes to transient devices, standard batteries just won’t cut it.
“Any device without a transient power source isn’t really transient,” said Montazami. “This is a battery with all the working components. It’s much more complex than our previous work with transient electronics.”
Previously Montazami worked on a proof-of-concept project that involved electronics printed on a single layer of a degradable polymer composite. The transient battery is made up of eight layers, including an anode, a cathode and the electrolyte separator, all wrapped up in two layers of a polyvinyl alcohol-based polymer.
The tiny battery, which is about 1 millimeter thick, 5 millimeters long and 6 millimeters wide, possesses the components, structure and electrochemical reactions that are all very close to commercial battery technology.
The only difference is that when you drop it in water, the polymer casing swells, breaks apart the electrodes and dissolves away. Though the battery does not yet completely disappear, it does contain nanoparticles that disperse as the battery’s casing breaks the electrodes apart.
For devices that will require larger batteries with higher capacities to provide more power, their batteries will take longer to self-destruct. According to the researchers’ paper, applications that require higher power levels could be connected to several smaller batteries.
Even though batteries are tried-and-tested technology, Montazami said the transient battery project presented three major challenges for his research group.
Though the team faced numerous challenges in the development of this first-run battery, Montazami says it remained dedicated since there are not many groups working on projects of this nature.