Glass, rubber and plastics are amorphous solids. While common, unlike other classes of solids, they have stymied researchers attempts to determine their atomic structures with precision. Mapping the 3D atomic structures of crystals, for example, is the impetus behind many advances in physics, chemistry, biology, materials science, geology, nanoscience and drug discovery. Amorphous solids lack rigid, repetitive atomic structures and therein lies the problem.
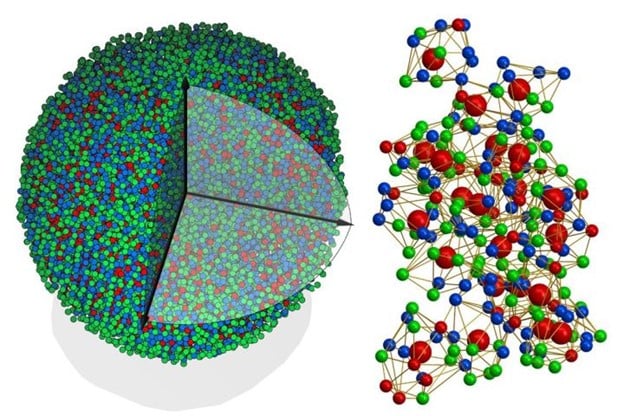
A recent study published in Nature by lead author, Jianwei “John” Miao, a UCLA professor of physics and astronomy and member of the California NanoSystems Institute at UCLA, reports on the first-ever determination of the 3D atomic structure of an amorphous solid–metallic glass. Metallic glasses are stronger and more shapeable than standard crystalline metals and have widespread use in such products as electrical transformers to high-end golf clubs and the housings for Apple laptops and other electronic devices.
Researchers used atomic electron tomography a type of 3D imaging pioneered by the team. The approach involves beaming electrons through a sample and collecting an image on the other side. When the sample is rotated, measurements can be taken from multiple angles, yielding data that is stitched together to produce a 3D image.
The researchers examined a sample of metallic glass about 8 nanometers in diameter, made of eight different metals. Using 55 atomic electron tomography images, they created a 3D map of the approximately 18,000 atoms that made up the nanoparticle. Although approximately 85% of the atoms were in a disordered arrangement, there were pockets where a fraction of atoms coalesced into ordered superclusters.
Original Source: Eureka Alert