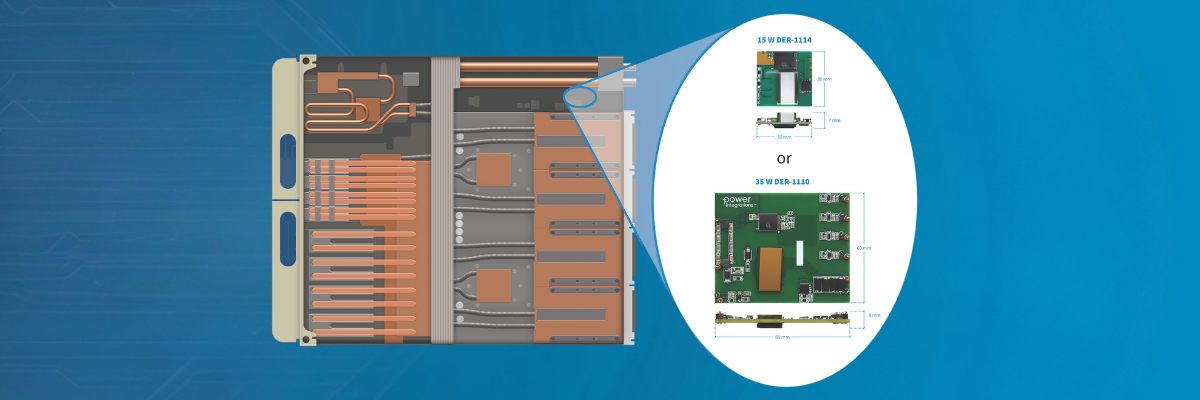
118 elements are categorized in the periodic table that we all studied in school. Each element is described with a one- or two-letter abbreviation, the number of protons that are in the element’s nucleus shown as the atomic number. The number of protons also determines the number of electrons that orbit the nucleus.
Recently, however, chemical engineers at EPFL’s School of Basic Sciences investigated another number that also needs to be reported, each element’s oxidation state, or oxidation number. This describes how many electrons an atom must gain or lose in order to form a chemical bond with another atom.
The oxidation state of a single element is straightforward, however when it comes to compounds made up of multiple elements, simple no longer exists. For complex materials, it is impossible to predict the oxidation state from first principles and most quantum programs require the oxidation state of the metal as input.
Predicting oxidation states now rests on something called “bond valence theory” that estimates the oxidation state of a compound based on the distances between the atoms of its constituent elements. This doesn’t always work, especially in materials with crystal structures. It is not only the distance that matters but also the geometry of a metal complex.
In a study, published in Nature Chemistry, researchers were able to train a machine-learning algorithm to categorize a famous group of materials, the metal-organic frameworks, by oxidation state. They used the Cambridge structural database, a repository of crystal structures in which the oxidation state in given in the name of the materials.
Researchers made a machine-learning model that has captured the collective knowledge of the chemistry community. By uploading a crystal structure and the machine-learned model, the results is the most likely oxidation state.
Original Release: Eureka Alert